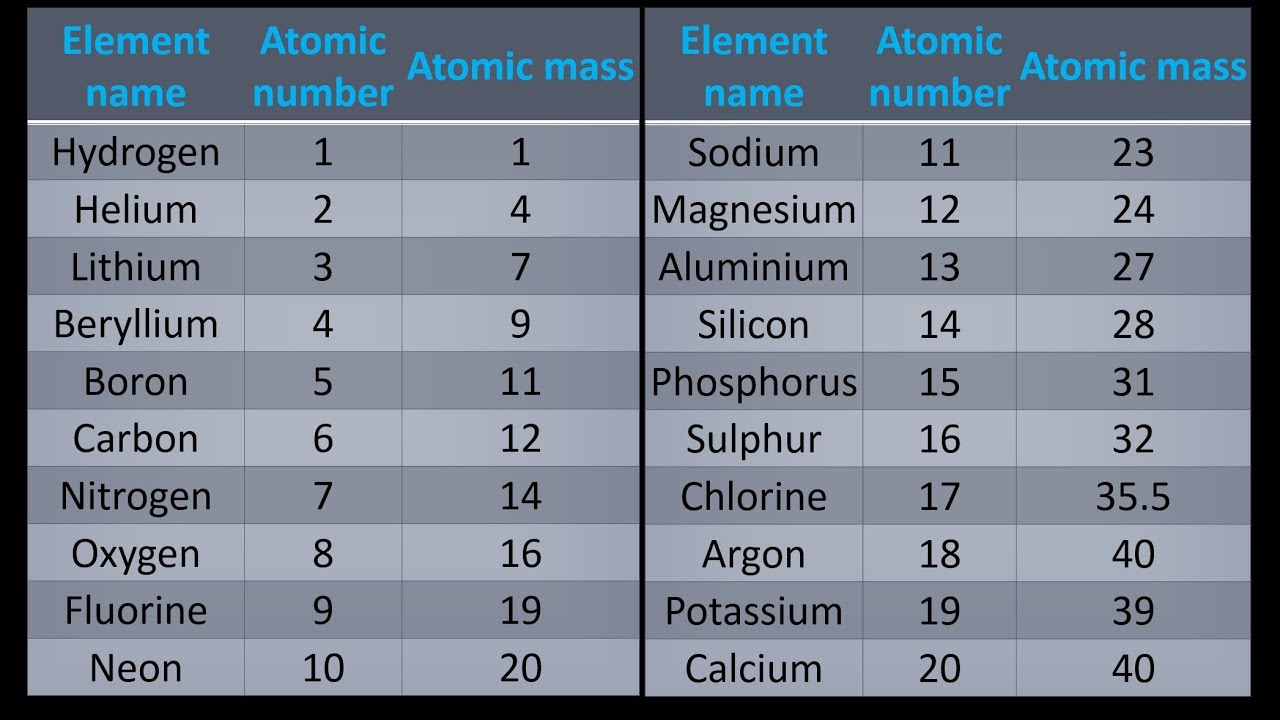
SHORE LEAVE Morton Salt maintains a plant near the south end of Utah's Great Salt Lake. It is an excellent disinfectant for swimming pools and water supplies, and its compounds are used in plastics such as polyvinyl chloride (PVC), stain removers, and bleach. Uses: Chlorine is essential to living systems and is also one of the top chemicals manufactured in the U.S. Breathing high concentration of the gas can be fatal chlorine was used as a poison gas during World War I. Today, most chlorine is produced through the electrolysis of aqueous sodium chloride.Īppearance: Yellowish-green, dense, sharp-smelling gas.īehavior: Liquid chlorine burns skin, and gaseous chlorine irritates mucous membranes. Occurrence: Found in nature dissolved in salts in seawater and in the deposits of salt mines. Identified as an element by Sir Humphry Davy in 1810. History: Discovered, yet misidentified as a compound, by Swedish chemist Carl Wilhelm Scheele in 1774. Name: From the Greek khloros, greenish yellow. In the small town of Javelle, France, chlorine added to alkaline water created l'eau Javelle ("bleach" in English, NaOCl in chemspeak) that was used in the fabric industry in the late-18th century. Sir Humphry Davy recognized the gas as an element in 1810 and named it based on the Greek word for its color, khloros.Ĭhlorine was by this time already in use. Carl Wilhem Scheele, a Swedish pharmacist, first described the greenish yellow gas in 1774 after dropping hydrochloric acid onto manganese dioxide. Sadly, the ≡ oxidation state of chlorine is often glossed over as the necessary counterion to an exotic metal ion or complex cation, the necessary yin to complement the yang.Ĭonversely, molecular chlorine, Cl 2, has held a starring role in history, both for its benefits to human health and for its detrimental effects on the environment. Arguably, chloride plays a more important role in the antitumor drug cisplatin, Pt(NH 3) 2Cl 2, although its function there is to get lost (that is, hydrolyze) so that platinum has room to bind to DNA. Sodium grabs all the press from NaCl as the dietary culprit in hypertension, while silver's the key to AgCl's action in photography. The molar mass of carbon dioxide is 12.01 + (2 × 16.00) = 44.01 g/mol.C&EN: IT'S ELEMENTAL: THE PERIODIC TABLE - CHLORINEĬHLORINE CYNTHIA BURROWS, UNIVERSITY OF UTAHĪ s an anion, chlorine is rather ordinary.CO 2 has one carbon atom and two oxygen atoms.Oxygen (O) has an atomic mass of about 16.00 amu.Carbon (C) has an atomic mass of about 12.01 amu.Let's calculate the molar mass of carbon dioxide (CO 2): Add them together: add the results from step 3 to get the total molar mass of the compound.Calculate molar mass of each element: multiply the atomic mass of each element by the number of atoms of that element in the compound.The atomic mass is usually found on the periodic table and is given in atomic mass units (amu). Find atomic masses: look up the atomic masses of each element present in the compound.For example, water is H 2O, meaning it contains two hydrogen atoms and one oxygen atom. Identify the compound: write down the chemical formula of the compound.
One mole contains exactly 6.022 ×10 23 particles (Avogadro's number) Mole is a standard scientific unit for measuring large quantities of very small entities such as atoms and molecules.Molar mass ( molar weight) is the mass of one mole of a substance and is expressed in g/mol.(1 u is equal to 1/12 the mass of one atom of carbon-12) Molecular mass ( molecular weight) is the mass of one molecule of a substance and is expressed in the unified atomic mass units (u).To calculate molecular weight of a chemical compound enter it's formula, specify its isotope mass number after each element in square brackets.Įxamples of molecular weight computations: Molar mass calculator also displays common compound name, Hill formula, elemental composition, mass percent composition, atomic percent compositions and allows to convert from weight to number of moles and vice versa.Ĭomputing molecular weight (molecular mass) Functional groups: D, Ph, Me, Et, Bu, AcAc, For, Tos, Bz, TMS, tBu, Bzl, Bn, DmgĮxamples of molar mass computations: NaCl, Ca(OH)2, K4, CuSO4*5H2O, nitric acid, potassium permanganate, ethanol, fructose, caffeine, water.Capitalize the first letter in chemical symbol and use lower case for the remaining letters: Ca, Fe, Mg, Mn, S, O, H, C, N, Na, K, Cl, Al. Sodium chloride appears as a white crystalline solid.Ĭomputing molar mass (molar weight)To calculate molar mass of a chemical compound enter its formula and click 'Compute'.
